atom
atomic structure
chemistry
physics
elements
subatomic particles
science education
Structure of Atom
Ode2Code
August 14, 2025
• 2 min read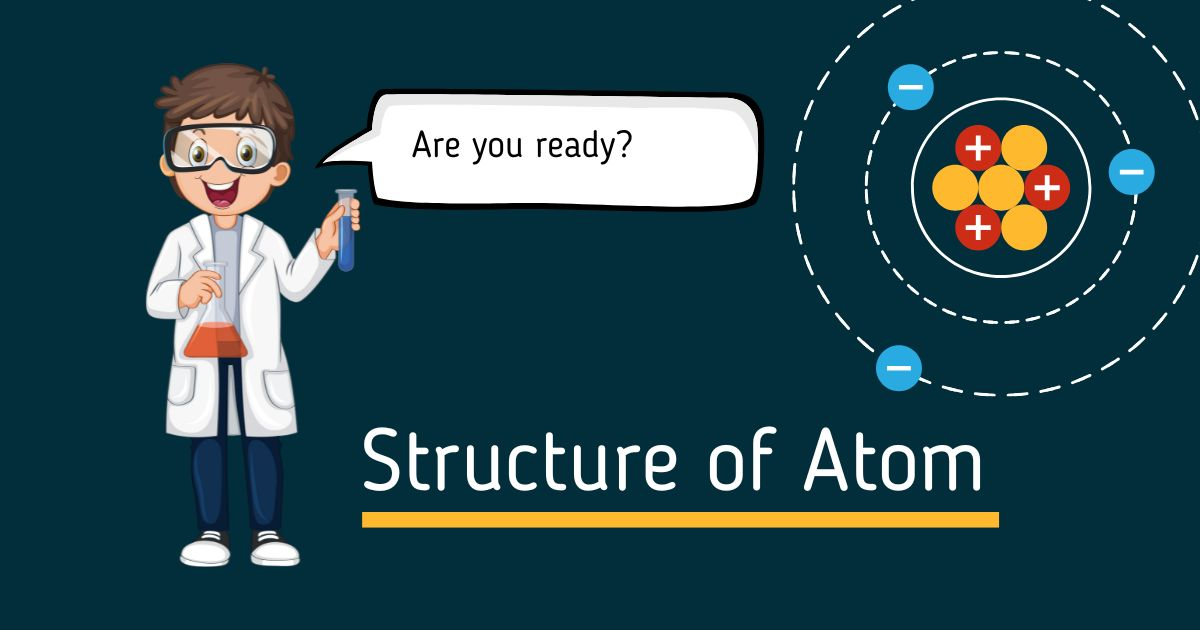
The structure of an atom is fundamental to grasping the principles of chemistry and physics. At its core, the nucleus houses positively charged protons and neutral neutrons. Surrounding this nucleus are negatively charged electrons that orbit in defined energy levels. This delicate balance of forces not only determines the atom's identity but also its chemical behavior, shaping the building blocks of matter.
The Nucleus: The Heart of the Atom
Let's break it down. The nucleus is incredibly small compared to the overall size of the atom, yet it contains most of the atom's mass. Here’s what you need to know:
- Protons: These subatomic particles carry a positive charge. The number of protons in the nucleus defines the chemical element. For example, hydrogen has one proton, while carbon has six.
- Neutrons: Neutrons are neutral particles, meaning they have no charge. They help stabilize the nucleus by providing a buffer between the repulsive forces of the positively charged protons.
The Electron Cloud: The Orbiting Particles
Now, surrounding the nucleus is the electron cloud. Electrons are much lighter than protons and neutrons and occupy regions of space defined by energy levels. Here’s what you need to know about electrons:
- Energy Levels: Electrons exist in specific energy levels or shells around the nucleus. These levels can be visualized as layers of an onion. The closer an electron is to the nucleus, the lower its energy.
- Electron Configuration: The arrangement of electrons in these energy levels determines how an atom interacts with others. For example, atoms with full outer shells are generally stable and less reactive.
Summary: The Balancing Act
What this really means is that the balance between protons, neutrons, and electrons defines everything about an atom. The nucleus creates a central core, while the electrons create a cloud of potential energy around it. This structure leads to the diverse properties of elements, from gases like oxygen to metals like gold.
Key Takeaway
Understanding the structure of an atom is crucial for anyone diving into science. It’s not just about knowing the names of particles; it’s about seeing how these tiny components interact to form the matter around us.
For further reading on atomic structure, feel free to check out more resources soon for a deeper dive.